 
Where m Higher energy level & n Lower energy level. The frequency of the emitted electromagnetic radiation results from the energy difference between the initial and the final state. The energy of an electron in an atom can only assume discrete values En. It also showed that his theory was only successful for hydrogen and closely related systems. However, it should be noted at this point that his third postulate (quantum condition) is no longer tenable from today’s perspective. BOHR’s postulatesīOHR solved the problem in 1913 by introducing postulates (casually: “by decree”) by transferring to the atom the quantization introduced by PLANCK for the black body and by EINSTEIN for the photon. However, since in RUTHERFORD’s atomic model all possible radii of the electron orbits and thus all-electron speeds were allowed, the total energy (potential energy + kinetic energy) of the electron cannot assume any discrete values. BALMER series reversal of the Na line FRANCK-HERTZ experiment), one must assume discrete energy states in the atom. Because of this experimentally proven fact (e.g. RUTHERFORD’s atomic model cannot explain the quantum emission and absorption of energy by the atoms. The consequence of this would be a crash of the electrons into the nucleus. From the classical point of view, the circling electrons execute an accelerated movement and accelerated charges emit electromagnetic energy. RUTHERFORD’s atomic model cannot explain the stability of the atoms. In some cases, BOHR’s postulates are not justified either, Roman Sexl speaks of a “legal solution “to the problems of the RUTHERFORD atomic model by the Dane Neil Bohr”. Notably, the retention of descriptive electron orbits around the nucleus has to be criticized from the point of view of quantum mechanics. He also found that the various energy levels can hold differing numbers of electrons: energy level 1 may hold up to 2 electrons, energy level 2 may hold up to 8 electrons, and so on.Bohr’s Atomic Model represents only an intermediate stage in the development of the model from the point of view of the quantum mechanical atomic model recognized today. The ground state of the atom was when the shells get closer and closer to one another, the further they are from the nucleus. He found that the closer an electron is to the nucleus, the less energy it needs, but the farther away it is, the more energy it needs. /Bohr-58e690203df78c51620ff02e.jpg)
Bohr called the collection of electrons that orbits at any distance an electron shell. While in their orbit, electrons do not lose any energy. The further they are from the nucleus, the greater amount of energy they possess. But from his special evidence, he concluded that electrons are found at only certain distances from the nucleus, and have particular values of energy. īohr agreed with Rutherford’s proposal that in the atom the electrons revolve around a central positively charged nucleus that is responsible for most of the weight of the atom. It was here when he proposed a new atomic model to explain the emission spectrum of hydrogen. 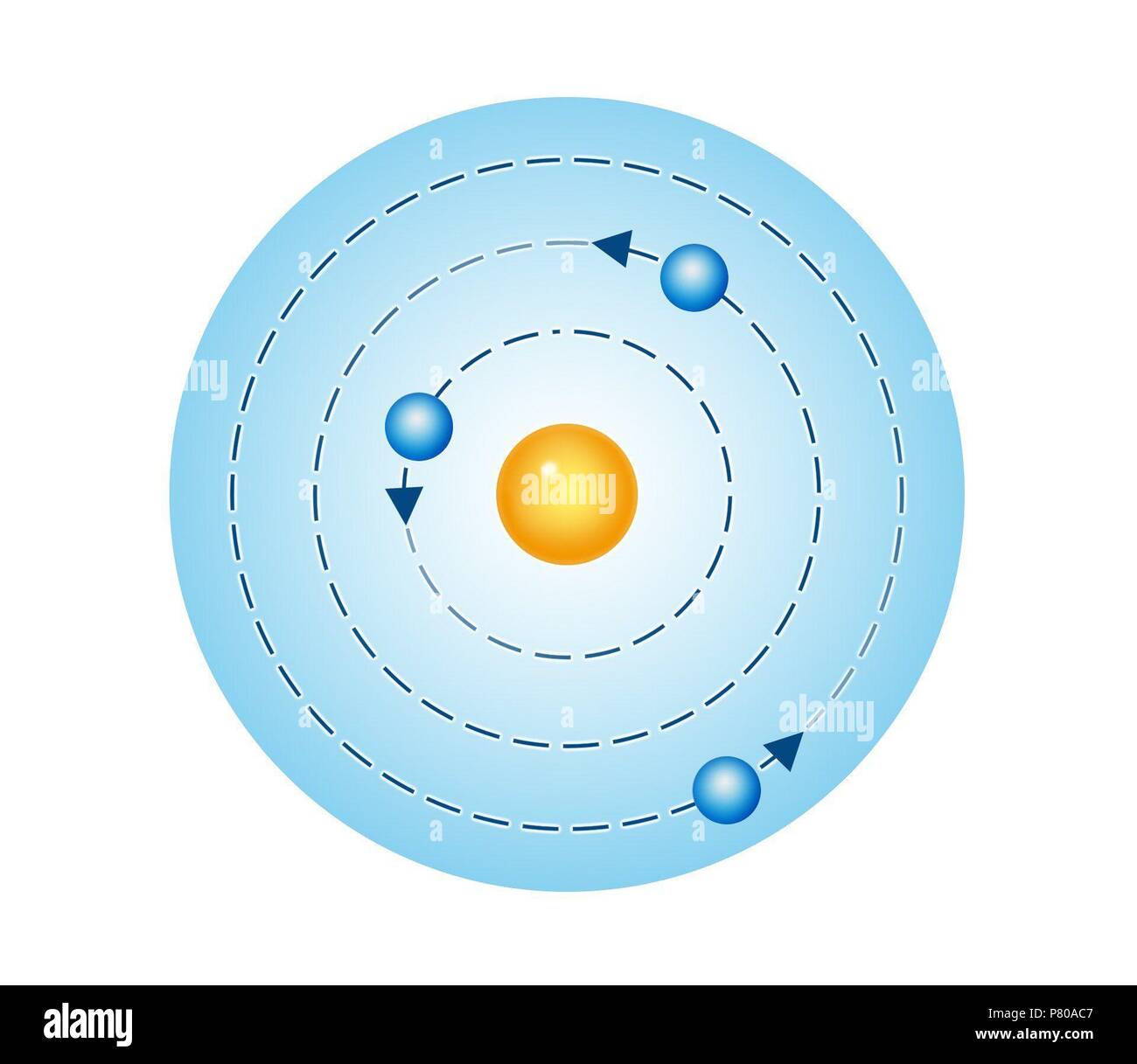
Bohr concluded that the laws of basic physics did not abide to objects on an atomic scale. Since the lines were not continuous like rainbow, the electrons were not giving of energy continuously. This meant that he observed the coloured lines (wavelengths given by a spectroscope). In 1913 Bohr analysed the spectral lines of hydrogen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
